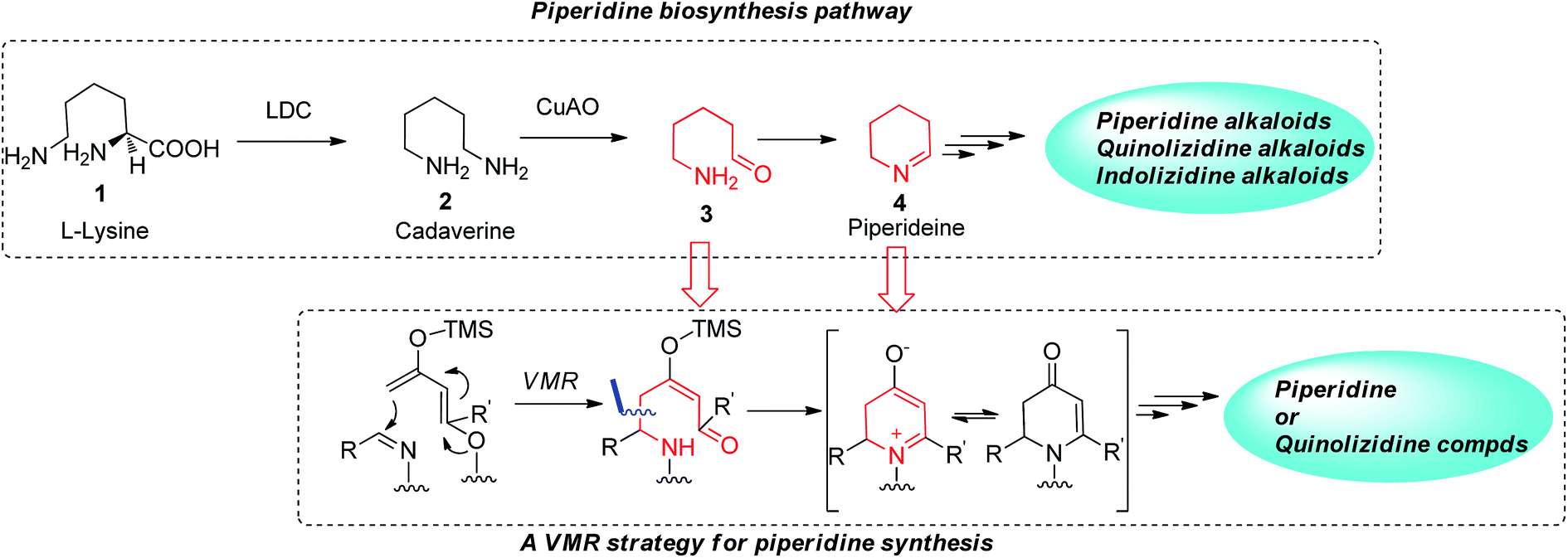
Building polyfunctional piperidines: a stereoselective strategy of a three-component Mannich reaction inspired by biosynthesis and applications in the ... - RSC Advances (RSC Publishing) DOI:10.1039/C4RA14418J
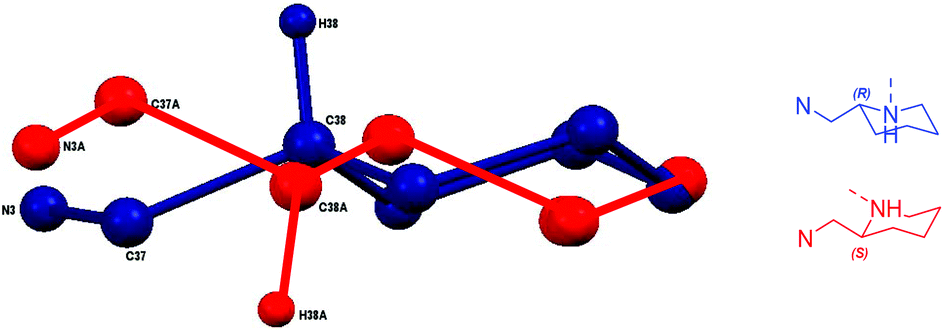
Ligands and complexes based on piperidine and their exploitation of the ring opening polymerisation of rac -lactide - Dalton Transactions (RSC Publishing) DOI:10.1039/C7DT00751E

A Mild and Efficient Procedure for Ring-Opening Reactions of Piperidine and Pyrrolidine Derivatives by Single Electron Transfer Photooxidation - ScienceDirect
Welcome to Chem Zipper.com......: Pyridine is almost 1 million times less basic than piperidine? Why?

Cyclisations and Strategies for Stereoselective Synthesis of Piperidine Iminosugars - Dhara - 2021 - The Chemical Record - Wiley Online Library
Piperidine nucleus in the field of drug discovery | Future Journal of Pharmaceutical Sciences | Full Text

organic chemistry - What is the expected number of rings obtained in the following ring formation process? - Chemistry Stack Exchange
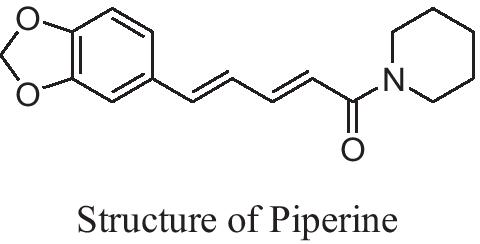
Piperidine nucleus in the field of drug discovery | Future Journal of Pharmaceutical Sciences | Full Text
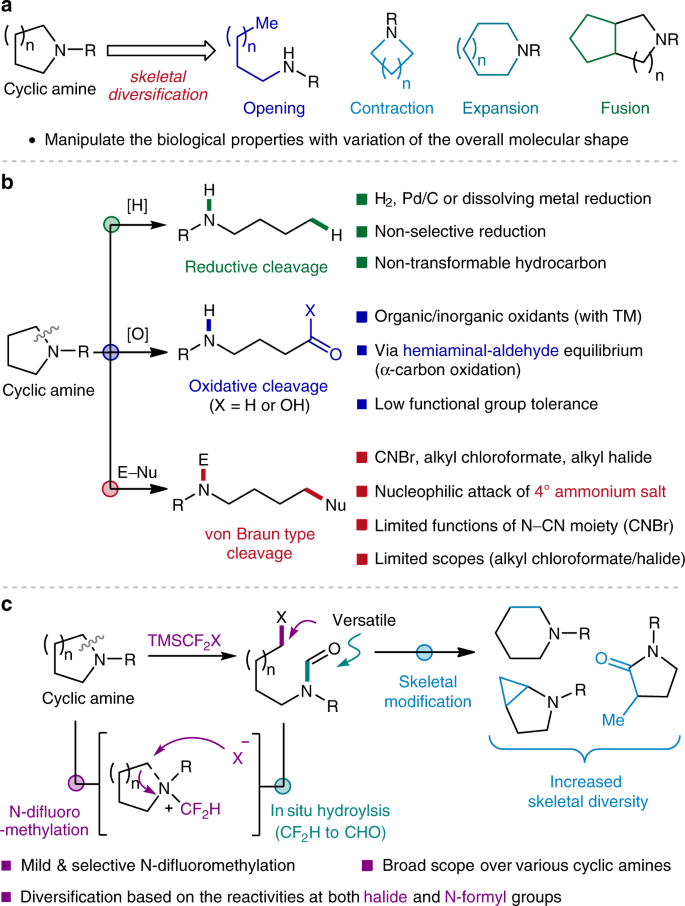
Ring-opening functionalizations of unstrained cyclic amines enabled by difluorocarbene transfer | Nature Communications

Plausible mechanism of 3,5-dispirosubstituted piperidine ring formation | Download Scientific Diagram
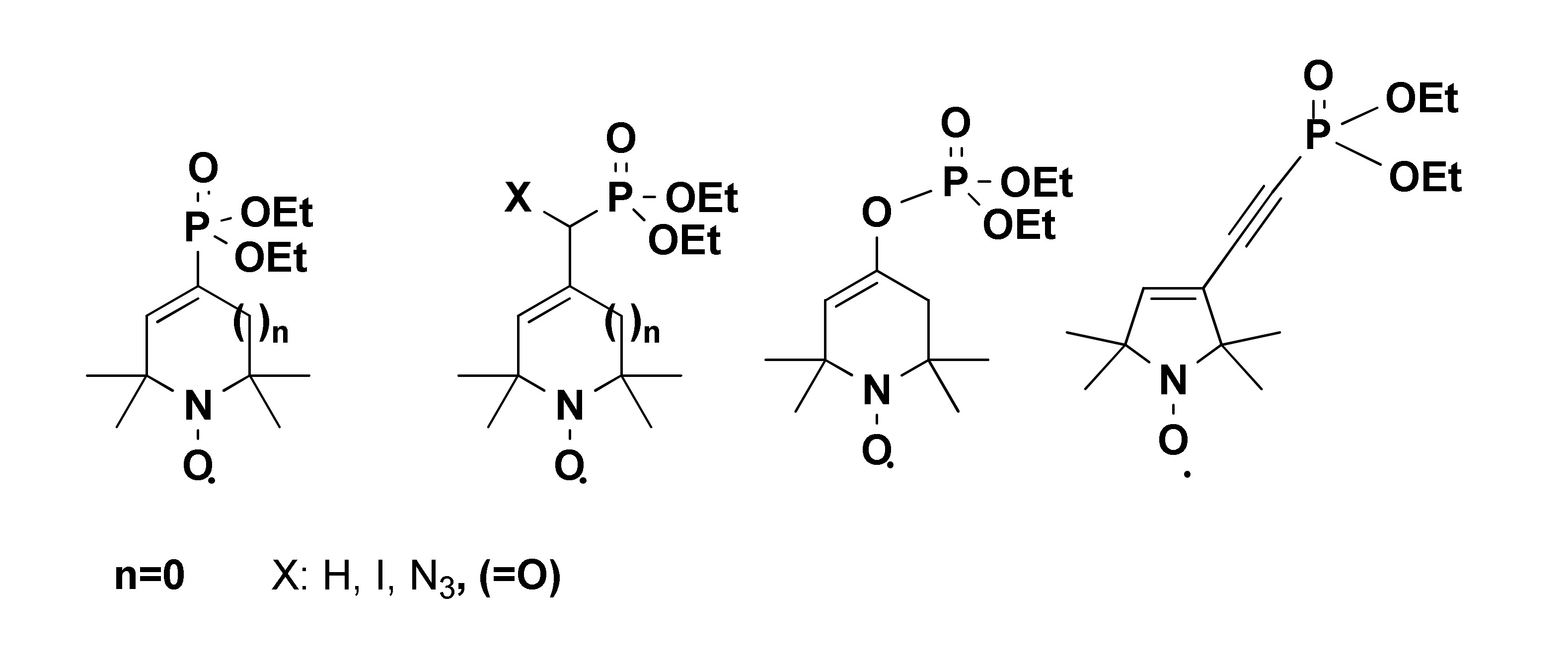
Molecules | Free Full-Text | Syntheses and Reactions of Pyrroline, Piperidine Nitroxide Phosphonates

Potent drugs with a bridged piperidine ring (3,4) and piperazine ring... | Download Scientific Diagram
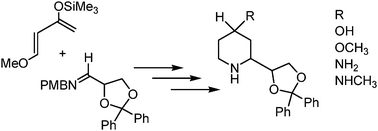
Synthesis and NMDA receptor affinity of dexoxadrol analogues with modifications in position 4 of the piperidine ring - MedChemComm (RSC Publishing)



![PDF] Use of Piperidine and Pyrrolidine in Knoevenagel Condensation | Semantic Scholar PDF] Use of Piperidine and Pyrrolidine in Knoevenagel Condensation | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/204d8cfb50b6267ae364d6e6a5a3077f018f4e9a/2-Figure2-1.png)